Flexiforming® Technology
One Catalyst. Many Feeds. Choice of Products.
Flexiforming is a zeolite-based catalytic process that converts naphtha, light olefins, alcohols, ethers, or their blends into high-octane gasoline, kerosene, or BTX aromatics. Operator-adjustable process lets you shift feed and product slate to match market conditions.
Unparalleled Feedstock Flexibility
Feed virtually any alcohol or ether, renewable or fossil naphtha, LPG, or light olefins. 6,000+ hours tested on 50+ feedstocks. Pick the cheapest combination available.
Proprietary HZSM-5 Catalyst
Modified zeolite provides high-temperature stability, steam resistance, and 3–5 year lifetime. Protected by 33 patents across 12 countries.
Adjustable Product Slate
Regulate temperature, pressure, and space velocity to shift output between gasoline, kerosene (SAF), and BTX in response to market conditions.
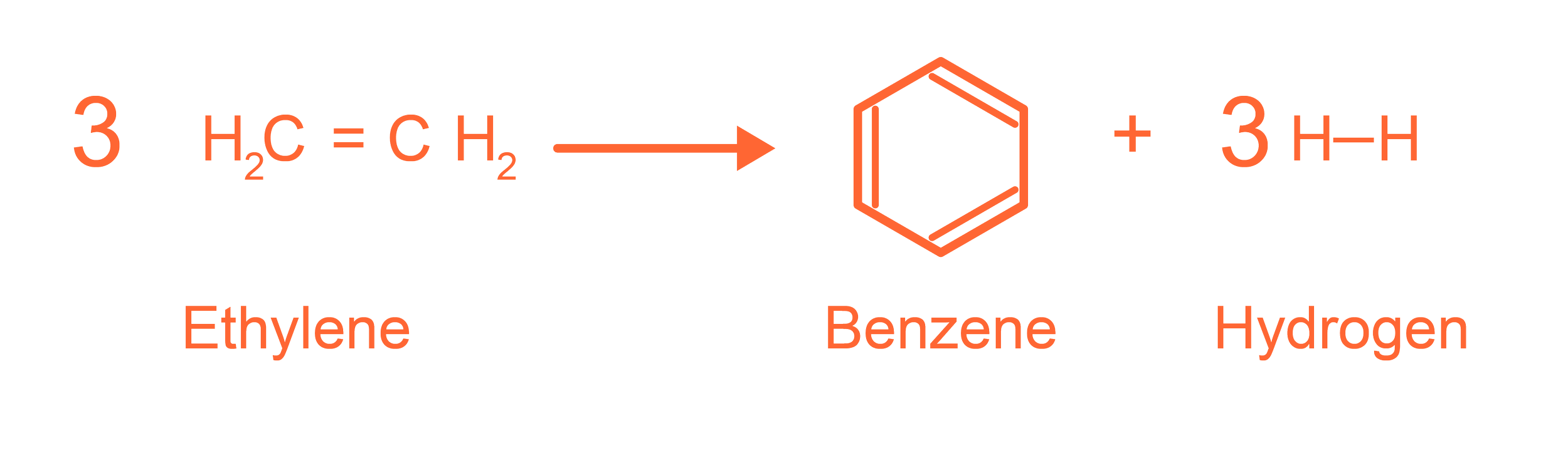
Key Reactions
Flexiforming Chemistry